Endeavour Awards for Projects Collaboration Using the Orca system at a University Setting
By Dr Vera Barbosa
The Endeavour Awards which were announced during the LEAP 4 event last month recognized the planning and hard work of young scientists doing their Masters’ degree project at University College London and Kings College London in collaboration with ProEconomy Ltd, who provided them with an Orca copper and silver ionisation system.
How does copper silver ionisation work?
- Copper and silver ionisation (CSI) involves the generation of copper and silver ions in water by water flowing through a turbine of a flow sensor.
- This sends a signal to the control unit, which then passes a low DC current between two copper and two silver electrodes located in an electrode chamber.
- The current causes ionisation, i.e. the release of copper and silver ions into the flowing water.
- Being electrically charged the copper and silver ions seek opposite polarity and find this in the negatively charged sites on cell wall of bacteria, such as Legionella, Pseudomonas and coli.
- The ions distort and weaken the cell wall and then damage the cell by binding at specific sites to DNA, RNA, cellular protein and respiratory enzymes denying all life support systems to the cell, causing death.
The system is a relatively new modality for the control of Legionella, Pseudomonas and other pathogens in water systems. The use of CSI was first recorded in the USA in 1990 (Lin et al. 2011), although it was pioneered by NASA in the 1960s (Albright et al. 1967). The ProEconomy CSI system is known by its trade name Orca.
What were the collaboration projects about?
The projects carried out at the labs at UCL investigated various aspects of the system including:
‘Inactivation of the bacterium Pseudomonas aeruginosa by CSI used for Legionella control’
‘Water discoloration and the staining of sinks where CSI is being used for Legionella and Pseudomonas control’
‘The tarnishing of sterilised medical instruments by water being treated with CSI for Legionella and Pseudomonas control’
‘Energy saving and carbon footprint reduction by reducing water temperature and using CSI for pathogens control in water systems.’
These projects results showed that:
- Medical instruments are typically made of austenitic stainless steel with strong corrosion resistance, however; tarnishing or corrosion of instruments is not an uncommon occurrence in hospitals (Brugirard, Guiraldenq & Mazille, 1991). There are claims that suggest that Cu and Ag ions in water can induce chemical interactions with stainless steel, leading to corrosion (Cooper et al. 1971). Therefore, this study aimed to determine the relationship between the CSI treatment and tarnishing of instruments. Tarnishing of medical equipment was not due to Cu or Ag ions but to the water softeners used. This phenomenon occurs in situations where Cl– concentration is high or halogenated softening salts have been used. The strong ionic pull of the softened water may cause pitting corrosion to occur. Pitting corrosion occurs when the passive layer of the stainless steel is removed by free Cl– in the water.
- Yellow and white stains were observed on the tiles surface after one week and became more apparent over time in both Orca and tap water while there was no obvious change in the tiles in deionised water. The white stains may be due to a mixture of the lime scale and trace silver chloride in the test sample, while in tap water the colour of stains may be attributed to the calcium carbonate mixed with sulphate ions and impurities. After a period of time grey-purple colouring might occur caused by photodecomposition and not due to silver ions.
- Control of high levels of aeruginosa was achieved with increasing levels of Cu and Ag ions. Figure 1 shows P. aeruginosa concentration (cfu/ml) at 0.6 mg/L Cu and 0.06 mg/L Ag, compared with the control tap water and deionised water. Figure 1 shows rapid increase in P. aeruginosa from 1.0 ×108 to 8.2 ×108 cfu/ml during 24 hours when the tap water was used instead of Orca water. Very little cell growth was observed in DI water as there was no carbon source.
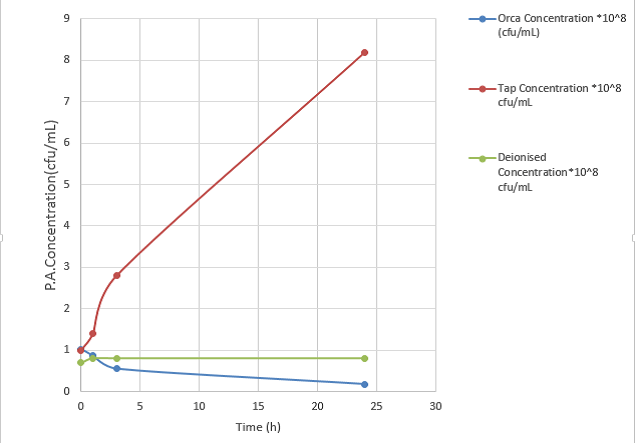
Figure 1: P. aeruginosa concentration versus time (Cu and Ag levels in water = 0.6/0.06 mg/L Cu/Ag; P. aeruginosa starting concentration in samples = 1.0 x 10-8)
- Energy calculations showed savings of 33% as a result of lowering water temperature in the premises. Figure 2 shows the savings in energy and carbon footprint.
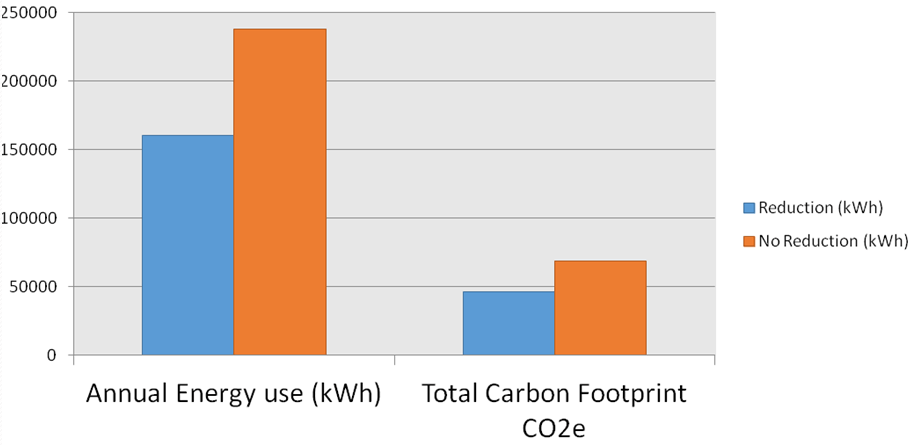
Figure 2: Annual energy use and total carbon footprint with reduction and without reduction in water temperature in the Morgan Stanley building.
The Endeavour Awards Prizes
1st prize – Choice of over 775 activities, including exclusive Champagne Balloon Flights for two, ultimate tailoring experience in London, luxury hotel Spa break for two, Tandem Skydives for four or an Ultimate Track Day, to a value of £1000. It was won by Leo (Zhibin Ma) from UCL who did a project on Pseudomonas control using the Orca CSI system.
2nd prize – Choice of over 600 experiences, including Rally Driving, Deluxe Spa Days, Country Pursuits, Thames Sightseeing and London Eye for Four, Recording Studio Session, Aerobatic Flights, Personal Shopping, Motor Racing and Eagle Handling. It was won by Diego Jimenez (UCL) and Daniel Wong (KCL), who did a joint project on energy saving using the Orca CSI system.
3rd prize – Choice from an array of over 400 interests, hobbies and activities including RIB power-boating on the Thames, supercar driving around a racetrack, hot air balloon options all over the countryside, or sushi-making classes for food lovers to a value of £100. It was won by Vanessa Onuogu from UCL who did a project on what causes medical equipment tarnishing.
References
Albright C.F., Nachum R., Lechtman M.D. (1967). Development of an electrolytic silver-ion generator for water sterilization in Apollo spacecraft water systems: Final Report. Apollo Applications Program, Manned Spacecraft Center, NASA:Houston. Available online: http://www.clearwaterpoolsystems.com/nasa-connection.html (Accessed May 2017).
Brugirard J., Guiraldenq P., Mazille H. (1988). Tarnishing and Corrosion of Semi-Precious Alloys for Dental Applications. Key Engineering Materials Vols. 20-28:1703-1709.
Cooper W., Jones, P. and Parfitt, G. (1971). Precipitation of silver chloride from homogeneous solution. Kolloid-Zeitschrift und Zeitschrift für Polymere, 246(2), pp.704-709
Lin Y., Stout J.E., Yu V.L. (2011). Controlling Legionella in Hospital Drinking Water: An Evidence-Based Review of Disinfection Methods. Infection Control and Hospital Epidemiology. 32(2):166-173.

